ICMCS researchers announce the invention of a new type of soft gel in the top journal ‘Science’
A new type of soft gel developed by School researchers may have important implications for the future use of liquid crystals.
Conventional wisdom says that particles and liquid crystals do not mix. In the latter state of matter, anisotropic molecules (which have direction-dependent properties) align to form various 'mesophases'. The simplest mesophase is the nematic, in which rod shape molecules all point in the same direction. The inclusion of particles disrupts such ordering, and gives rise to energetically costly 'defects' known as disclinations. In the past, therefore, it has proved all but impossible to coax anything more than a handful of particles to disperse in liquid crystalline mesophases.
Now, researchers from the School's Institute of Condensed Matter and Complex Systems have managed to persuade the nematic phase of a well-known liquid crystal to mix with a high concentration of particles. The resulting material is a solid gel. 'Solidifying' liquid crystals this way may have important implications for their application. Liquid crystals are already widely used in display devices, and are increasingly seen as possible biomedical sensors. But liquid crystals flow like liquids, and so hitherto, there is always the need for an external container. The invention of liquid crystalline gels removes this constraint.
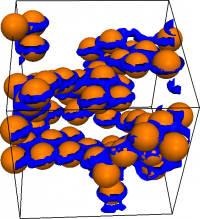
Extensive computer simulations allowed the ICMCS researchers to show that the new liquid crystal gel they had made in the test tube derived its rigidity from the disclinations entangling each other and trapping a percolated particle network (see figure, where particles are orange spheres, and disclinates are blue lines). Interestingly, this arrested state of matter has similarities with 'vortex glasses' in Type II superconductors, and may open up new avenues for understanding 'glassy arrest', which remains one of the deepest problems in condensed matter science.

